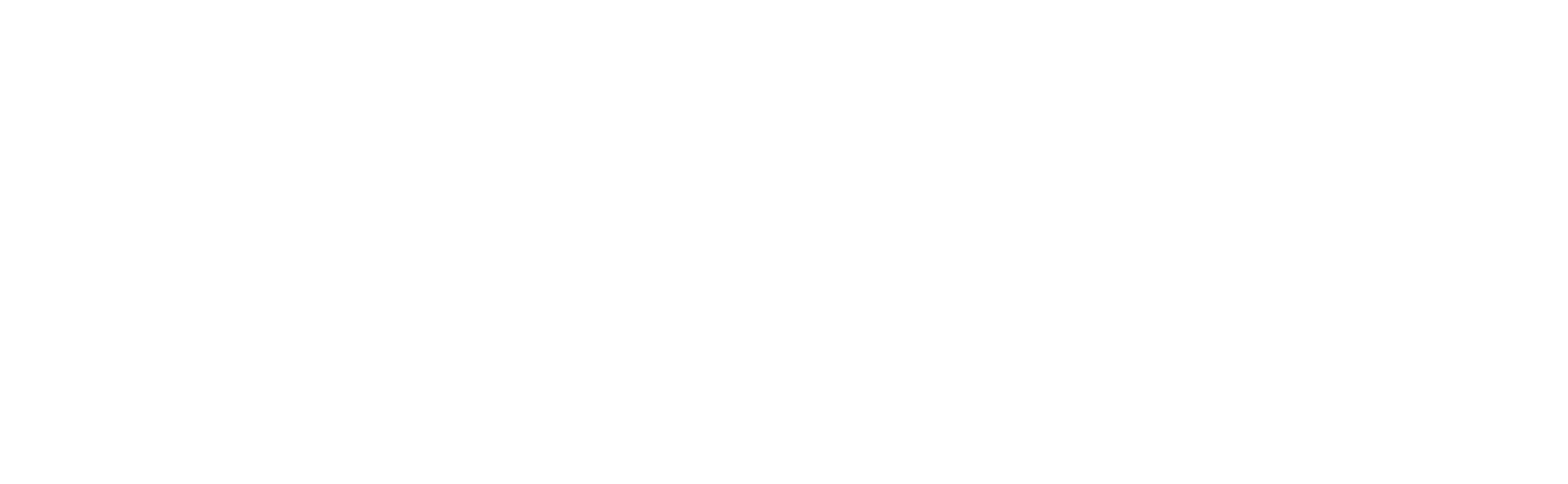July 11, 2019 : 3 min read
FDA 2579 Form: What Is It And Where Do I Get One?
I get calls every week from people at imaging facilities saying in panicked voices, “My physicist is here and he is looking for some form that I am supposed to have; some kind of FDA Form?” Does this sound familiar to you?
I'm sure the mere presence of the initials “FDA” is the main reason there is a trace of panic in the person's voice, but the situation isn't as dire as it may seem. Don’t get me wrong, the FDA 2579 form is a very important form and definitely has to be filled out and kept on record. It is, however, very easy to obtain.
Let me answer the 2579 questions I get most often so that you can have the documentation you need, when you need it.
What is an FDA 2579 form?
One of the many responsibilities of the Food and Drug Administration (FDA) is to regulate X-ray emitting medical imaging equipment. Under the Department of Health and Human Services, the FDA requires that any time a piece of X-ray emitting medical imaging equipment is installed an FDA 2579 form is filled out by the installing engineer or assembler. Essentially, by filling out the form the installer is saying they installed the device per manufacturer instructions.
Please note that the need for a 2579 is not limited to the installation of a complete system. There are a number of components which, if installed on a separate occasion such as a repair or a field upgrade, will also need to have a 2579 filed. To see which parts fall in this category, click the linked sample image above or the download button at the end and refer to section 4.
In many cases, the installing engineer must be registered with the local state’s radiological and/or health department to work on X-ray equipment. Any reputable organization working with medical X-ray systems should be familiar with this form. If you want to know and understand your state’s compliance requirements concerning “assemblers” visit your state’s radiology health department website. You might also benefit from checking out our post on whether or not you need lead lining for your X-ray room or you can download a list of radiological and health departments links to learn more about state requirements.
Where can I get the FDA 2579 form?
The engineer responsible for installing your X-ray imaging system should bring the FDA 2579 form with them when they install your equipment. If they do not have one and cannot get one, you will need to order it from the FDA. You can do this by visiting this link and following the instructions - FDA Request of 2579 Forms. You could also contact Block Imaging’s project management team who can assist with obtaining one.
If you already have a 2579 on file but have misplaced your onsite copy, take a look at this post to find out how you can remedy that: What to Do If You Can't Find your 2579
How Is an FDA 2579 Submitted?
The engineer should send the original and three carbon copies to the following locations:
- White Original – Mail to the FDA
- Yellow copy – Mail to State Radiology Health Department
- Pink copy – Left onsite for you, the equipment owner
- Blue copy - Remains with the installing engineer or “assembler”
Please keep in mind that the 2579 form is an original document with an assigned number. That number is assigned to that site and equipment, so you can’t make copies of one form to use for others. When the engineer performs an installation, they are verifying that a specific device is operating within the manufacturer’s specifications and attest to this by filling out and submitting the FDA 2579 form.
Why Do I Have to Have a Copy?
Beyond the obvious, surface-level answer that it's the law and your physicist will want to see it, there are a couple of other reasons to keep your 2579 on file.
- FDA compliance is often a part of the accreditation process.
- If there should ever be an incident involving your equipment and a patient injury, your 2579 is a valuable part of demonstrating that, to your best knowledge, your equipment was installed properly.
If the company you're working with isn't familiar with this form take caution and consider contacting Block Imaging's Project Management Team who can connect you with one of our qualified and approved engineers to get your form filled out and your facility in compliance.

Danny Fisher
Danny Fisher is a Project Manager at Block Imaging. His goal is to understand the unique needs of each project and deliver a detail-oriented execution. When he's not in the office, Danny enjoys spending time with his wife, catching up on the latest movies/TV, and staying in the know on the latest tech gadgets. He also loves finding great deals and inexpensive ways to travel the world